Hydrogen in welding is controlled by reducing diffusible hydrogen, limiting tensile and residual stress, and preventing crack-sensitive microstructures from forming in the weld area. In practical terms, this requires the use of low-hydrogen consumables, proper storage and handling, clean joint preparation, and controlled thermal practices such as preheat and interpass temperature management.
This issue is especially important because hydrogen cracking often appears after welding has been completed rather than during the weld itself. That delayed cracking can result in rework, inspection failures, production delays, and reliability concerns in structural, pipeline, and other critical applications.
What Is Hydrogen Cracking in Welding?
Hydrogen cracking is a weld defect that occurs when hydrogen remains in the weld region and combines with tensile stress and a crack-sensitive microstructure. It is commonly referred to as hydrogen-assisted cracking, cold cracking, or delayed cracking, depending on the material and welding context.
This form of cracking typically occurs in the weld metal or the heat-affected zone. Because it may develop hours after welding, it is particularly difficult to manage in production environments where the weld may initially appear sound during visual inspection.
For that reason, hydrogen cracking is not simply a defect identification issue. It is a process control issue that must be addressed before, during, and sometimes after welding.
Why Is Hydrogen a Problem in Welding?
Hydrogen becomes a welding problem when it remains in the joint long enough to interact with stress and a susceptible metallurgical structure. The risk is especially high in high-strength steel, thicker sections, restrained joints, and applications where cooling rates are rapid and residual stress is difficult to relieve.
In many cases, the concern is not just hydrogen alone, but hydrogen acting together with unfavorable welding conditions. A weld may look acceptable immediately after fabrication and still develop cracks later if the procedure does not sufficiently control the underlying risk factors.
This is why hydrogen control welding practices are essential in industries where weld integrity, service life, and safety performance cannot be left to chance.
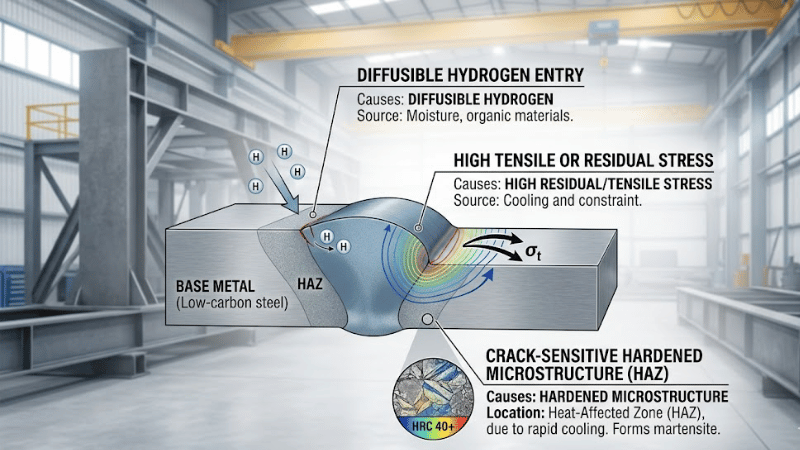
What Causes Hydrogen Cracking in Welding?
Hydrogen cracking in welding usually requires three conditions to be present at the same time: diffusible hydrogen, tensile or residual stress, and a crack-sensitive microstructure. When these three factors combine, cracking risk rises significantly.
The first condition is the presence of hydrogen. This can come from moisture in electrodes, fluxes, or welding wire, or from contamination on the base material. The second condition is the formation of a hard or brittle microstructure during cooling, which is more susceptible to crack initiation. The third condition is stress in the joint, often created by restraint, weld shrinkage, section thickness, or joint design.
This three-condition model is useful because it explains why how to prevent hydrogen cracking cannot be reduced to a single action. Controlling hydrogen requires attention to consumables, cleanliness, thermal input, cooling behavior, and joint restraint as part of one coordinated procedure.
To quickly assess your hydrogen risk and apply the correct metallurgical safeguards, refer to the iKratz Engineering Standards for hydrogen control below:
Quick Reference: Hydrogen Control & Crack Prevention
| Risk Factor | Why It Increases Cracking Risk | How to Reduce It (ikratz Standards) |
|---|---|---|
| Moisture in Consumables | Adds diffusible hydrogen directly into the molten weld pool. | Store in ovens; rebake per manufacturer (H-grade filler). |
| Dirty Base Material | Oil, rust, and paint decompose under arc heat to release H2. | Clean 50mm (2″) zone with stainless brush/solvent. |
| Low or No Preheat | Increases cooling rate, trapping H2 in brittle martensite. | Critical: Mandatory if CEIIW is 0.41-0.43. |
| Poor Interpass Control | Inconsistent thermal cycles lead to localized hardness spikes. | Maintain temperature; ensure t8/5 is 10-25 seconds. |
| High Joint Restraint | Raises residual and tensile stress across the HAZ. | Improve joint design; apply post-heat/stress relief. |
| Wrong Process Choice | Processes like SMAW (non-low H) increase hydrogen input. | Use GTAW for roots or ultra-low-H GMAW/FCAW. |
Where Does Hydrogen in Welding Come From?
Hydrogen in welding usually comes from moisture, contamination, poor consumable handling, or process conditions that increase the level of diffusible hydrogen in the weld zone. Identifying the source is one of the first steps in reducing cracking risk.
Moisture in consumables is one of the most common causes. Electrodes, fluxes, and some welding wires can absorb moisture when packaging is left open too long or when storage conditions are not properly controlled. Once that moisture is exposed to the heat of the arc, hydrogen can enter the weld metal.
Surface contamination is another major source. Oil, rust, paint, coatings, dirt, and moisture on the base material can all contribute to hydrogen pickup. Even seemingly minor contamination can undermine an otherwise well-designed procedure if it is present in the weld area.
Hydrogen can also be introduced through poor handling discipline. Wet gloves, contaminated tools, and uncontrolled exposure of consumables in field conditions may appear minor at first, but they often become contributing factors in delayed cracking problems.
How Do You Control Hydrogen in Welding?
Does Preheat Reduce Hydrogen Cracking?
Yes, preheat can significantly reduce hydrogen cracking risk. It does so by slowing the rate of cooling, lowering the likelihood of hard microstructure formation, and allowing more time for hydrogen to diffuse out of the weld area.
Preheat is especially valuable in high-strength steels, thicker sections, and restrained joints where cracking conditions are more severe. In these cases, correct preheat practice can play a major role in improving weld reliability and reducing the chance of delayed cracking.
However, preheat should not be treated as a complete solution on its own. It cannot compensate for wet consumables, contaminated surfaces, or an unsuitable welding process. The best outcomes are achieved when preheat is integrated into a broader hydrogen control procedure.

Which Welding Practices Reduce Hydrogen Risk Most?
The welding practices that reduce hydrogen risk most effectively are straightforward, but they must be applied consistently. Use low-hydrogen consumables, store them correctly, clean the weld area thoroughly, apply the right preheat, and maintain proper interpass temperature throughout the weld sequence.
It is also important to select the welding process according to the material, joint design, and service conditions. In addition, reducing unnecessary restraint where possible can help lower the stress component that contributes to cracking.
Where required, post-weld thermal treatment should also be considered. In practice, hydrogen cracking is often the result of several moderate control failures occurring together rather than one dramatic procedural mistake.
Best Practices for Different Applications
Hydrogen control requirements vary according to material strength, joint restraint, section thickness, and welding environment. The fundamental principles remain consistent, but the level of control required changes with the application.
For high-strength steel, tighter process discipline is essential because the material is more likely to form crack-sensitive microstructures. Low-hydrogen procedures, carefully selected preheat, and consistent interpass temperature control are especially important in these cases.
For pipeline welding and field welding, environmental exposure adds another layer of complexity. Moisture, variable handling conditions, and inconsistent heat control can increase risk, so consumable protection and procedural discipline become even more critical.
For thick sections and highly restrained joints, residual stress is higher and thermal balance is harder to maintain. In these applications, hydrogen control must be built into procedure development from the beginning rather than applied as a troubleshooting measure after cracking occurs.
For critical fabrication environments, repeatability also matters. A procedure that works only under ideal shop conditions may not provide the consistency needed for demanding production or field service requirements.
Common Mistakes That Increase Hydrogen Cracking Risk
Many hydrogen-related weld failures are caused by several small mistakes working together rather than one obvious error. That is one reason why the problem can be difficult to identify if the procedure has not been reviewed carefully.
Common causes include wet or poorly stored consumables, dirty or damp joint surfaces, inadequate preheat, poor interpass temperature control, excessive restraint, and incorrect assumptions about material behavior. Another frequent mistake is relying on a low-hydrogen filler metal alone while neglecting storage, cleaning, and thermal management.
Process selection errors can also increase risk. A welding method that does not suit the material, thickness, or jobsite conditions may create unnecessary variability and increase hydrogen-related cracking potential.
Effective hydrogen control in welding depends on consistent procedural discipline. When control weakens across several steps, cracking risk increases even if no single mistake appears severe on its own.
FAQs
What is hydrogen cracking in welding?
Hydrogen cracking is a weld defect caused by the combination of diffusible hydrogen, tensile stress, and a crack-sensitive microstructure. It often appears after welding rather than during welding, which is why it is also called delayed cracking.
What causes hydrogen cracking?
Hydrogen cracking usually occurs when three conditions exist together: hydrogen is present, the weld area forms a susceptible microstructure, and the joint contains enough tensile or residual stress to initiate cracking.
Does preheat reduce hydrogen cracking?
Yes. Preheat helps reduce hydrogen cracking by slowing cooling, reducing hardness, and giving hydrogen more time to diffuse away from the weld area.
Is hydrogen cracking more common in high-strength steel?
Yes, generally. High-strength steel is more susceptible because it can form crack-sensitive microstructures more easily, especially in thicker sections and restrained weldments.
Conclusion
Controlling hydrogen in welding means controlling the full welding procedure rather than focusing on one isolated variable. Hydrogen cracking develops when hydrogen, stress, and a susceptible microstructure are allowed to exist together, so prevention depends on coordinated decisions in consumable selection, storage, cleanliness, preheat, and process control.
The most reliable results come from disciplined, repeatable welding practices that reduce cracking risk before the weld is completed. When hydrogen control welding is approached systematically, manufacturers can improve weld quality, reduce rework, and achieve more stable performance across demanding applications.
Need Help Choosing the Right Hydrogen Control Method?
Choosing the right hydrogen-control approach depends on the base material, joint design, welding process, environment, and production requirements. With strong technical support and application-based guidance, it becomes much easier to reduce cracking risk, improve consistency, and avoid costly corrective work.
Contact us today to discuss your welding application, evaluate suitable low-hydrogen solutions, and identify the most reliable approach for preventing hydrogen cracking in your process.




