Hydrogen is usually not “corrosive” in the way most people mean. It typically does not eat away at metal like acids or oxidizing chemicals do. The bigger concern is hydrogen embrittlement—when hydrogen enters certain metals and makes them less ductile, less tough, and more likely to crack.
That is why the better question is not just “is hydrogen corrosive?” but whether hydrogen can damage a specific material under real operating conditions.
Quick Answer
Is hydrogen corrosive?
Usually not in the normal chemical-corrosion sense. In most hydrogen systems, the bigger risk is hydrogen embrittlement or hydrogen-assisted cracking.
Can stainless steel be used in hydrogen service?
Often yes, but it depends on grade, microstructure, strength, weld condition, cold work, pressure, temperature, and loading.
When is general guidance not enough?
High-pressure, cyclic, welded, cold-worked, or safety-critical applications usually need deeper engineering review instead of rule-of-thumb selection.
What Is Hydrogen Embrittlement, and How Is It Different From Corrosion?
Plain-English definition
Hydrogen embrittlement happens when hydrogen enters a metal and reduces ductility and toughness. A part may still look normal, but it can become more crack-prone under stress.
Why it is different from ordinary corrosion
Corrosion is usually chemical attack at the surface. Embrittlement changes mechanical behavior inside the material and may not leave obvious visual signs.
Terms readers should not confuse
- Hydrogen embrittlement: loss of ductility or toughness due to hydrogen
- Hydrogen-induced cracking: cracking associated with hydrogen
- Stress corrosion cracking: cracking from stress plus a corrosive environment
- General corrosion: normal chemical attack and material loss
| Critical Factor | General Corrosion | Hydrogen Embrittlement (HE) |
|---|---|---|
| Primary Mechanism | Chemical/Electrochemical attack | Atomic hydrogen diffusion & lattice stress |
| Visual Indicators | Rust, pitting, wall-thinning | None (often invisible internal cracks) |
| Detection Method | Visual, UT (thickness check) | Destructive testing, specialized NDT |
| Engineering Threshold | Corrosion allowance (mm/yr) | Hardness < 22 HRC / Ni ≥ 12% |
| Impact of Welding | Weld decay, oxidation | HAZ sensitization & residual stress |
While standard inspections are a baseline, understanding the limitations of [RT vs UT testing] is crucial, as microscopic hydrogen-induced cracks often fall below the detection threshold of conventional radiography
Is Hydrogen Corrosive to Metal?
Why “corrosive” is often the wrong question
Most users really mean, “Can hydrogen damage metal?” That is the more useful question. Hydrogen may not be strongly corrosive chemically, but it can still create serious mechanical integrity risks.
When hydrogen creates real material risk
Hydrogen damage risk increases with high pressure, high stress, cyclic loading, long service life, and susceptible alloys.
A better screening question
Ask whether hydrogen will reduce toughness, increase cracking risk, or shorten fatigue life in the actual material and service condition.

Can Stainless Steel Suffer Hydrogen Embrittlement?
Why stainless steel is not one answer
Hydrogen embrittlement stainless steel behavior varies by family. Austenitic, martensitic, duplex, and precipitation-hardening stainless steels do not respond the same way.
Why austenitic stainless steels are often preferred
Many austenitic stainless steels are often more resistant than high-strength or hardened steels, which is why they are widely considered for hydrogen service.
When stainless steel can still fail
Cold work, poor weld condition, stress concentration, high pressure, and cyclic service can still raise failure risk. Stainless steel is not automatically safe just because it is stainless.
What engineers should check
Review:
- Grade
- Microstructure
- Strength or hardness
- Fabrication history
- Pressure and temperature
Which Materials Are Most Vulnerable to Hydrogen Damage?
Higher-risk materials
High-strength steels, hardened steels, and heavily cold-worked steels are generally more vulnerable.
Lower-risk but not risk-free options
Some austenitic stainless steels, selected nickel alloys, and other hydrogen-suitable materials may perform better, but only when matched to the actual service conditions.
Key takeaway
Material selection is a balance of strength, toughness, fabrication condition, and operating environment.
Hydrogen Gas vs Hydrogen Peroxide: What Is the Difference?
Why these are separate questions
Hydrogen gas is mainly a mechanical integrity issue. Hydrogen peroxide is mainly a chemical compatibility and corrosion issue. They should not be treated as the same material-selection problem.
When peroxide should be handled separately
If the application involves concentration, storage, cleaning, or oxidizer compatibility, hydrogen peroxide needs its own evaluation.
What Factors Actually Determine Real-World Risk?
Pressure and temperature
Higher pressure can increase hydrogen-related risk. Temperature can also affect diffusion, toughness, and cracking behavior.Safety protocols and pressure limits should adhere to the global best practices outlined by the European Industrial Gases Association (EIGA), particularly regarding carbon steel and stainless steel transitions.
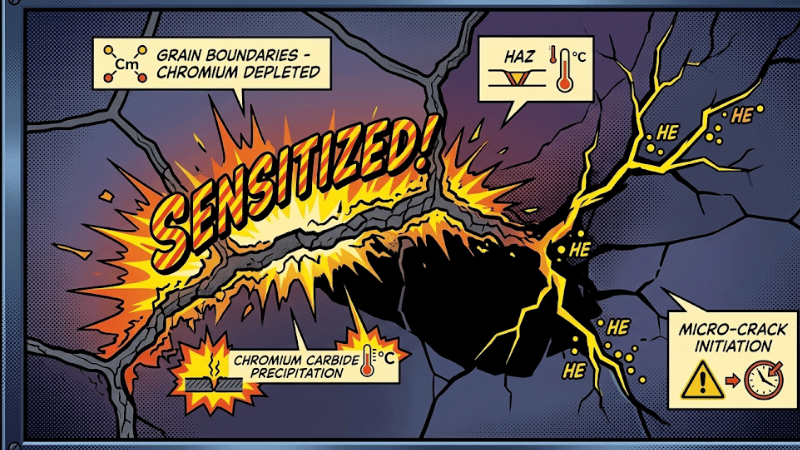
Expert Insight: The environmental baseline during fabrication is a critical yet frequently ignored variable. I recall a high-stakes project in New Delhi in June 2007, where ambient temperatures regularly hit 45°C. This extreme heat significantly altered the cooling physics of the manual TIG welds.
Because the pipes couldn’t dissipate heat quickly enough, the material lingered too long within the 450°C–850°C sensitization range, leading to chromium carbide precipitation at the grain boundaries. When combined with the minor alignment tolerances inherent in manual welding, these microscopic “weak spots” couldn’t handle the pressure.
We were forced to scrap the entire system after micro-cracks appeared during the final integrity test. This experience is exactly why we now prioritize automated orbital systems with environmental compensation; in the world of high-pressure gas, the weather can be just as dangerous as the weld parameters.
Stress and cyclic loading
Cyclic loading is especially important because it can accelerate crack initiation and growth.
Material condition and weld history
Cold work, hardening, weld heat-affected zones, and residual stress can significantly change performance.
Purity, contaminants, and service environment
Real service conditions matter. Contaminants or mixed environments can change the damage mechanism and severity.
How Should You Choose Materials for Hydrogen Service?
Step 1: Define exposure conditions
Start with pressure, temperature, purity, duty cycle, and environment.
Step 2: Define consequence of failure
Consider safety, leakage risk, downtime, and inspection difficulty.
Step 3: Screen the material and condition
Check alloy family, hardness, product form, fabrication route, and weld condition.
Step 4: Decide whether validation is needed
High-consequence systems should not rely on general rules alone. They often need standards review, vendor data, or validation testing.
Before finalizing any international project, cross-reference local regulatory requirements using the HyLaw Database to ensure your material selection meets both technical and legal standards.
Common Mistakes in Hydrogen Material Selection
Assuming stainless steel is always safe
It is not. Grade and condition matter.
Treating embrittlement like ordinary corrosion
A corrosion-only mindset can miss the real failure mechanism.
Ignoring cold work, welding, or hardness
These factors can strongly affect hydrogen compatibility.
Choosing strength before compatibility
Higher strength often brings higher hydrogen sensitivity.
Skipping validation in high-risk systems
That is a common shortcut—and a costly one.
When Should Stainless Steel Selection Go to Engineering Review?
A practical checklist
Escalate to engineering review when you need to confirm:
- Exact alloy
- Product form
- Stress level
- Welded or cold-worked condition
- Pressure and temperature
- Applicable standards or vendor guidance
Pass, caution, or validate further
A simple low-risk case may pass screening. A welded, cold-worked, or high-pressure case usually deserves caution. A safety-critical system often needs formal validation.
Conclusion
Final answer in plain English
Hydrogen is usually not corrosive in the ordinary sense. The bigger concern is hydrogen embrittlement and cracking risk. Stainless steel can work in hydrogen service, but the right answer depends on grade and service conditions.
The decision logic to remember
Do not ask only, “Is hydrogen corrosive?” Ask whether hydrogen can damage this specific material under actual operating conditions.
FAQs
Is hydrogen corrosive?
Usually not in the standard chemical-corrosion sense. In most cases, the bigger concern is whether it increases cracking risk.
Can stainless steel suffer hydrogen embrittlement?
Yes. Some stainless steels perform better than high-strength steels, but grade, cold work, weld condition, and service conditions still matter.
Should hydrogen peroxide and stainless steel be covered in the same guide?
CTA
Usually no Hydrogen peroxide is a chemical compatibility issue, while hydrogen gas is mainly a hydrogen embrittlement issue.
Don’t let a rule-of-thumb material choice turn into a million-dollar failure—from the high-pressure labs of Vienna to the 45°C heat of New Delhi, we’ve learned that hydrogen integrity is won or lost in the details of the weld. Contact the iKratz technical team today for a professional review of your hydrogen piping specifications and orbital welding protocols.




